Search

The Kids researchers have been awarded over $3.4 million for a new trial to pioneer improved ways for managing cystic fibrosis (‘CF’).

In a WA first, researchers from The Kids Research Institute Australia have shown that Aboriginal babies are 22.5 times more likely to be treated for skin infections than non-Aboriginal babies.

A new study has confirmed the changing pattern of meningococcal disease in Western Australia.

Join us for stories, imagination and connection in the Discovery Centre

Get ready for a week of mind-blowing fun as the Discovery Centre comes alive for National Science Week with this year’s exciting theme: Decoding the Universe!

A powerful screening of truth telling that captures the stories of the Western Australia Stolen Generation.

Find out what our communities had to say about the draft Statement on Consumers and Community Involvement in Health and Medical Research.

The Wesfarmers Centre of Vaccines & Infectious Diseases brings together a number of independent researchers and research teams with a common aim; to find and deliver new and improved solutions to prevent and treat serious infections experienced by children or adolescents.
At the Wesfarmers Centre, we undertake research in five key areas of infections and immunisation to assist in children's health.
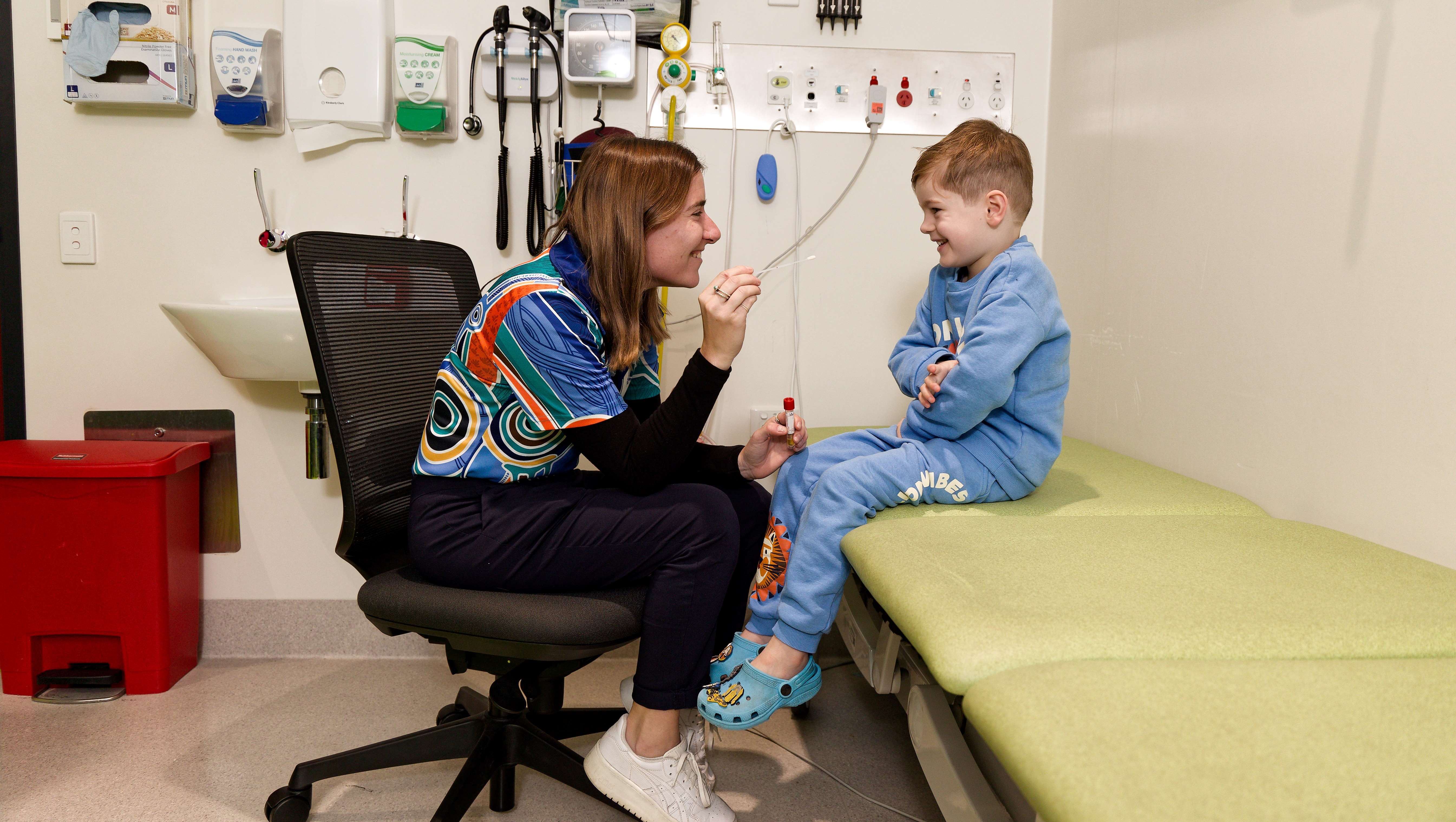
Researchers from The Kids Research Institute Australia would like to understand more about respiratory syncytial virus (RSV) and how we can provide the best protection for kids.
